In the News
AI Beats Physicians After-Visit Summaries for Hospital Patients
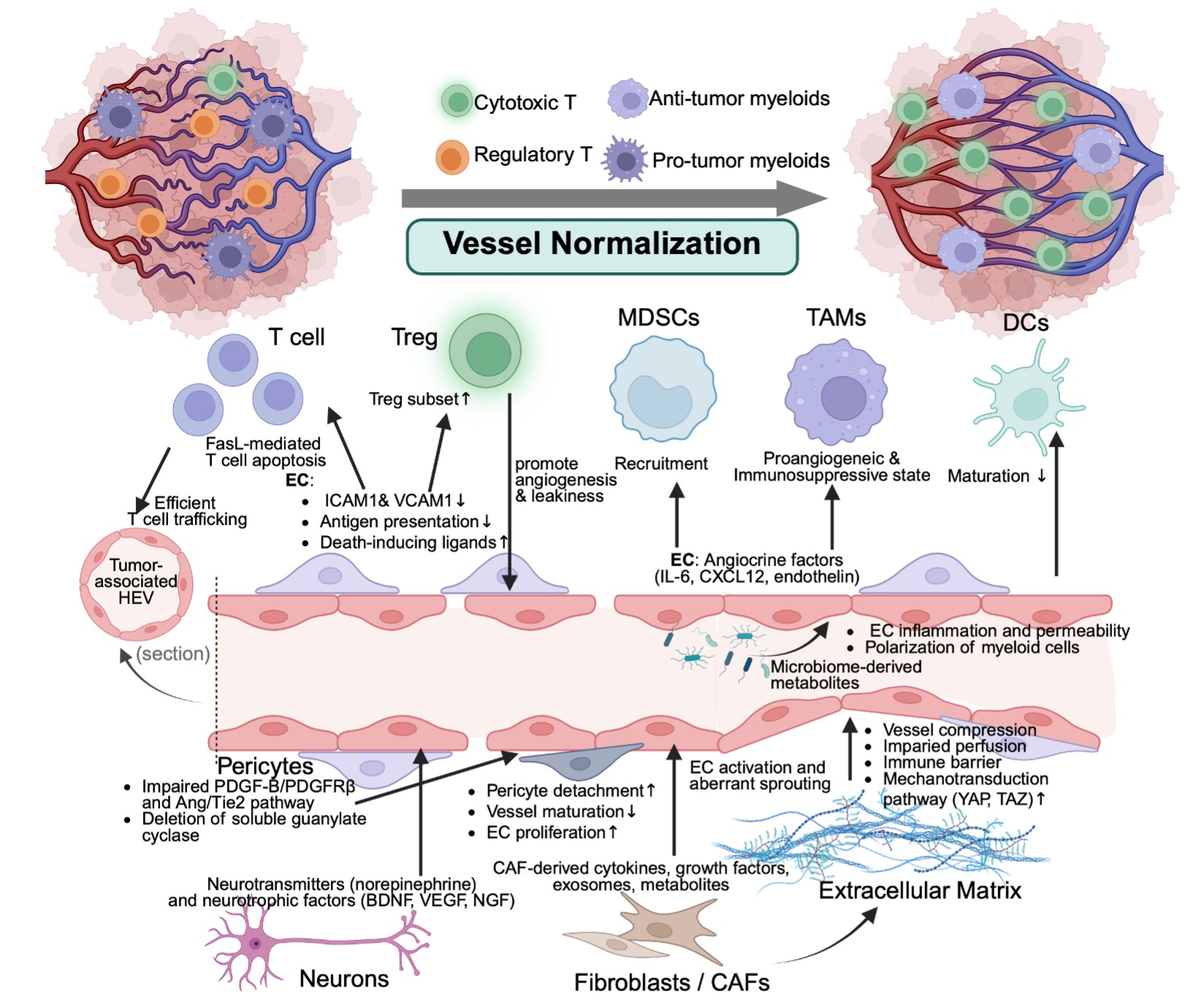
Over the past 25 years, anti-angiogenic therapies have moved from a simple "starve the tumor" strategy to a far more nuanced understanding of blood vessels as an active and reprogrammable component of tumors. Here, we elucidate this evolution by asking not just how tumors build abnormal blood vessels, but how those vessels shape the immune microenvironment, resist therapy, and can be strategically “normalized” to improve patient outcomes. We also wanted to capture an emerging frontier: how factors beyond classic growth factors, such as, the nervous system, gut microbiome, sex hormones, aging, and exercise, can also influence tumor vasculature in ways that could open entirely new therapeutic avenues.


