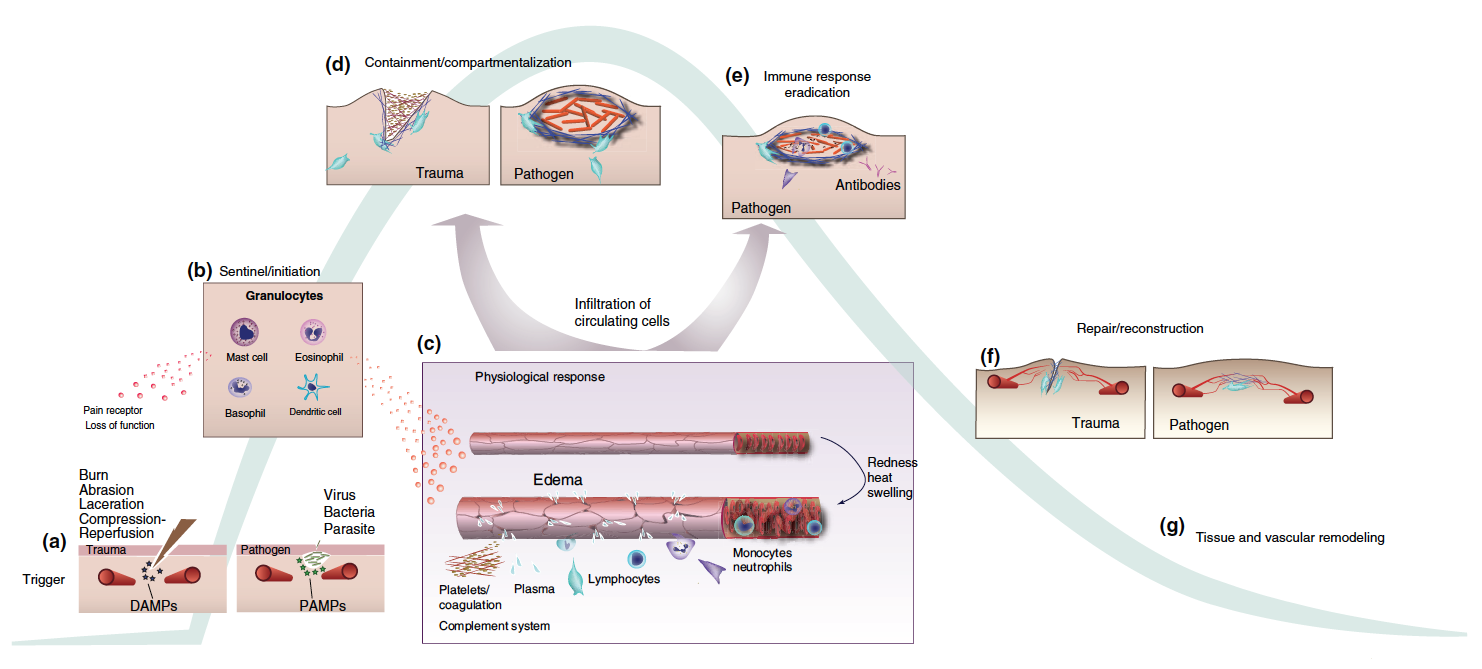
In the News
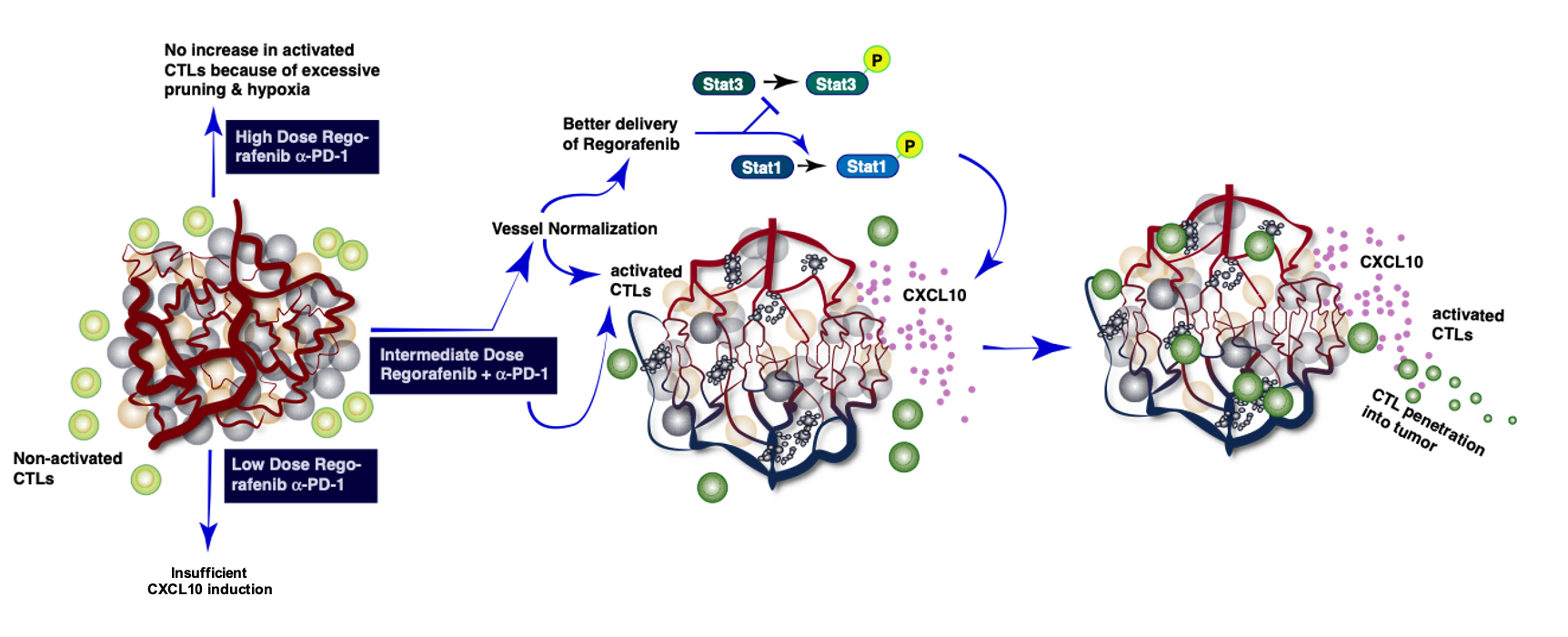
Judiciously dosed regorafenib combined with PD1 blockade increases CD8 T-cell infiltration by inducing CXCL10 expression in hepatocellular carcinoma (HCC).

Melding biology and physical sciences yields deeper understanding of cancer
BOSTON – An evolving understanding of cancer that incorporates the physical properties of tumors and their surrounding tissues into existing biologic and genetic models can direct cancer researchers down previously uncharted avenues, potentially leading to new drugs and new treatment strategies, say investigators from Massachusetts General Hospital (MGH), Harvard Medical School (HMS) and the Ludwig Center at HMS.
Chen IX, Newcomer K, Pauken K, Juneja VR, Naxerova K, Wu MW, Pinter M, Singer M, Sharpe AH, Jain RK
A bilateral tumor model identifies transcriptional programs associated with patient response to immune checkpoint blockade
Natl Acad Sci U S A. 2020;:ePub - PMID: 32907939
DOI: 10.1073/pnas.2002806117
Immune checkpoint inhibitors can effectively boost patients’ immune systems to fight cancer, but many do not benefit from these medications. A team led by Ludwig Harvard investigator Rakesh Jain reported in the Proceedings of the National Academy of Sciences a preclinical study describing an approach to identify potential clinical markers that indicate which patients will respond to such therapies and which might benefit more from others.
Celebrating the 30th anniversary of Rakesh Jain as Director of the Steele Laboratories
Their applications were chosen from among many excellent proposals considered by the review committee. This award will provide an opportunity to give a lecture/seminar at a national or international institution, organized by the Center for Faculty Development (CFD).
In addition, they have been pre-selected to participate in the Leadership Development Program for Researchers, which is also sponsored by the CFD. This course aims to prepare investigators for challenges inherent in establishing and maintaining a successful research program.
MGH will be recognizing the 2020 class of Anne Klibanski Postdoctoral Scholars at the Celebration of Science on September 21.
Brain tumors store and exert solid mechanical forces both internally and on the surrounding brain tissue. Here we sought to answer the question: can we decouple the physical effects from the biological effects of a brain tumor? The technique is described in a study published in Nature Protocols
It’s currently difficult to screen for certain liver diseases and to monitor these conditions once they’re discovered. A team led by investigators at Massachusetts General Hospital (MGH) and the Massachusetts Institute of Technology (MIT) recently developed a non-invasive imaging method that has promising clinical potential to accomplish both goals. The technique is described in a study published in Nature Biomedical Engineering.
Dan G. Duda, DMD, PhD, Director of Translational Research in GI Radiation Oncology, Department of Radiation Oncology, has been inducted into the American Institute for Medical and Biological Engineering (AIMBE) College of Fellows. Election is among the highest professional distinctions accorded to a medical and biological engineer. The College of Fellows is comprised of the top 2% of medical and biological engineers.
Normalizing the tumor microenvironment could help powerful immune-enhancing drugs better reach their targets and work more effectively, mathematical models predict
Cancer-associated fibroblasts (CAFs) are a key component of the tumour microenvironment with diverse functions, including matrix deposition and remodelling, extensive reciprocal signalling interactions with cancer cells and crosstalk with infiltrating leukocytes. As such, they are a potential target for optimizing therapeutic strategies against cancer. However, many challenges are present in ongoing attempts to modulate CAFs for therapeutic benefit. These include limitations in our understanding of the origin of CAFs and heterogeneity in CAF function, with it being desirable to retain some antitumorigenic functions. On the basis of a meeting of experts in the field of CAF biology, we summarize in this Consensus Statement our current knowledge and present a framework for advancing our understanding of this critical cell type within the tumour microenvironment